Tuesday Poster Session
Category: Liver
P3940 - Ashwagandha-Induced Liver Injury
Tuesday, October 24, 2023
10:30 AM - 4:00 PM PT
Location: Exhibit Hall

Has Audio

Sheena Bhushan, MD
Northeast Georgia Medical Center
Atlanta, GA
Presenting Author(s)
Sheena Bhushan, MD, Jawad Ilyas, MD, MS
Northeast Georgia Medical Center, Atlanta, GA
Introduction: Drug induced liver injury (DILI) is one of the leading causes of acute liver failure within the United States. While several classes of drugs are known to cause DILI, a recent uptick in herbal and dietary supplement induced DILI has been observed, accounting for 9% of all cases. Here the authors present a case of DILI in a 28-year-old due to an Ashwagandha herbal supplement.
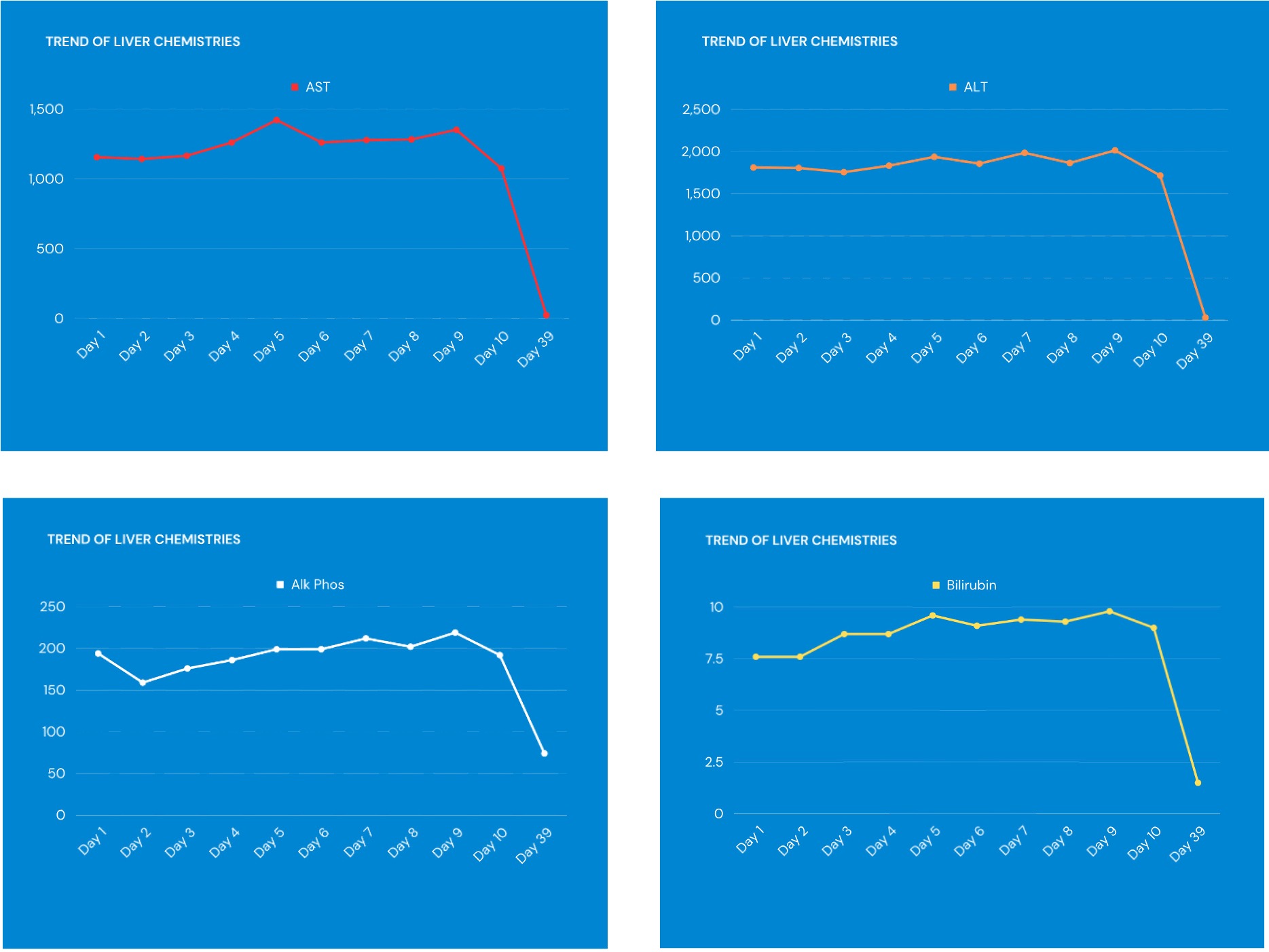
Case Description/Methods: A 28-year-old male with a past medical history of hypertension presented to the emergency department with generalized itching, worsening malaise, and jaundice. He denied any fever, changes in bowel habits, alcohol or recreational drug abuse. He also denied any changes in medications, except for starting ashwagandha for his anxiety about two months ago before bed. On admission vitals were unremarkable. Physical exam was remarkable for jaundice and did not show any hepatosplenomegaly, ascites, encephalopathy, or other stigmata of chronic liver disease. On arrival, pertinent laboratory work included a hemoglobin of 14.7 g/dl, white blood cell count of 4.0 k/uL, platelets of 212, AST 1157 u/L, ALT 1811 u/L, Total bilirubin 7.60 mg/dL, alkaline phosphatase 194 u/L and INR 0.97. His R-factor was calculated to be >5, consistent with a hepatocellular pattern. Hepatitis panel, autoimmune panel, and acetaminophen levels in blood were negative. Right upper quadrant ultrasound was unremarkable. CT scan of the abdomen and pelvis was unremarkable for any acute abnormalities. The patient was admitted to the hospital, and started on N-acetylcysteine, and supportive care. On discharge, he was advised to discontinue ashwagandha; however other medications before admission were restarted. A follow up visit in clinic 39 days after his hospitalization showed normalization of liver chemistries (figure 1) and resolution of his symptoms.
Discussion: Ashwagandha (Withania somnifera )is a popular Ayurvedic herbal supplement propagated as an anxiolytic, antidiabetic agent, antidepressant, and sleep aid. Specifics on its safety profile, adverse effects, and hepatoxicity are largely unknown. The liver injury manifests 2-12 weeks after initiating the supplement and typically has a cholestatic or mixed pattern of injury. Since herbal supplements are considered food substances, they are largely unregulated by the FDA. Further research in this area at a larger scale is warranted.
Disclosures:
Sheena Bhushan, MD, Jawad Ilyas, MD, MS. P3940 - Ashwagandha-Induced Liver Injury, ACG 2023 Annual Scientific Meeting Abstracts. Vancouver, BC, Canada: American College of Gastroenterology.
Northeast Georgia Medical Center, Atlanta, GA
Introduction: Drug induced liver injury (DILI) is one of the leading causes of acute liver failure within the United States. While several classes of drugs are known to cause DILI, a recent uptick in herbal and dietary supplement induced DILI has been observed, accounting for 9% of all cases. Here the authors present a case of DILI in a 28-year-old due to an Ashwagandha herbal supplement.
Case Description/Methods: A 28-year-old male with a past medical history of hypertension presented to the emergency department with generalized itching, worsening malaise, and jaundice. He denied any fever, changes in bowel habits, alcohol or recreational drug abuse. He also denied any changes in medications, except for starting ashwagandha for his anxiety about two months ago before bed. On admission vitals were unremarkable. Physical exam was remarkable for jaundice and did not show any hepatosplenomegaly, ascites, encephalopathy, or other stigmata of chronic liver disease. On arrival, pertinent laboratory work included a hemoglobin of 14.7 g/dl, white blood cell count of 4.0 k/uL, platelets of 212, AST 1157 u/L, ALT 1811 u/L, Total bilirubin 7.60 mg/dL, alkaline phosphatase 194 u/L and INR 0.97. His R-factor was calculated to be >5, consistent with a hepatocellular pattern. Hepatitis panel, autoimmune panel, and acetaminophen levels in blood were negative. Right upper quadrant ultrasound was unremarkable. CT scan of the abdomen and pelvis was unremarkable for any acute abnormalities. The patient was admitted to the hospital, and started on N-acetylcysteine, and supportive care. On discharge, he was advised to discontinue ashwagandha; however other medications before admission were restarted. A follow up visit in clinic 39 days after his hospitalization showed normalization of liver chemistries (figure 1) and resolution of his symptoms.
Discussion: Ashwagandha (Withania somnifera )is a popular Ayurvedic herbal supplement propagated as an anxiolytic, antidiabetic agent, antidepressant, and sleep aid. Specifics on its safety profile, adverse effects, and hepatoxicity are largely unknown. The liver injury manifests 2-12 weeks after initiating the supplement and typically has a cholestatic or mixed pattern of injury. Since herbal supplements are considered food substances, they are largely unregulated by the FDA. Further research in this area at a larger scale is warranted.

Figure: Figure 1: Trend of liver chemistries
Disclosures:
Sheena Bhushan indicated no relevant financial relationships.
Jawad Ilyas indicated no relevant financial relationships.
Sheena Bhushan, MD, Jawad Ilyas, MD, MS. P3940 - Ashwagandha-Induced Liver Injury, ACG 2023 Annual Scientific Meeting Abstracts. Vancouver, BC, Canada: American College of Gastroenterology.
