Monday Poster Session
Category: IBD
P2250 - Multiple Myeloma in a Patient With a Severe Ulcerative Colitis Exacerbation on Ustekinumab
Monday, October 23, 2023
10:30 AM - 4:15 PM PT
Location: Exhibit Hall
Has Audio

Ethan M. Cohen, MD
West Virginia University School of Medicine
Morgantown, WV
Presenting Author(s)
Ethan M.. Cohen, MD1, Ayowumi A. Adekolu, MD1, Jennifer Hadam-Veverka, MD2
1West Virginia University School of Medicine, Morgantown, WV; 2West Virginia University, Morgantown, WV
Introduction: Inflammatory bowel disease (IBD) is a term that includes two chronic inflammatory diseases of the GI tract, Crohn's disease and ulcerative colitis (UC). Uncontrolled inflammation can lead to bleeding, abdominal pain, hospitalization, need for surgery, and malignancy. Over the last decade, several new medications called biologics and small molecules have revolutionized the course of IBD by controlling inflammation and achieving remission. These medications come with risks which can include immunosuppression, infections or even malignancies. A detailed discussion regarding the risks and benefits of therapy should be tailored for each patient prior to starting therapy. In addition, ongoing monitoring for possible medication side effects and active disease should be done regularly.
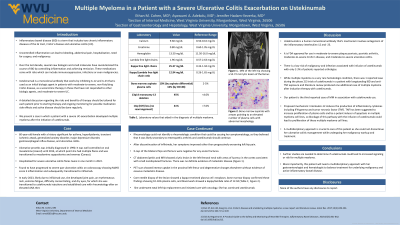
Case Description/Methods: 60-year-old female with a history of UC diagnosed in 1990 in remission and not on medical therapy was admitted for a severe acute UC flare. Her colonoscopy showed MAYO 3 inflammation and biopsies were negative for cytomegalovirus (CMV). She improved on IV steroids and was transitioned to infliximab (IFX) as an outpatient with a steroid taper. After induction with IFX, she developed severe side effects including a psoriatic rash along with significant joint pain. Given the side effects, she was switched to ustekinumab (UST). After her first infusion with UST, her joint pain worsened, and she developed limited mobility in her left hip. A CT scan showed a lytic lesion in the left femoral neck suspicious for malignancy, and a biopsy confirmed multiple myeloma (MM). She is now undergoing treatment for her multiple myeloma but will need medications to address her active UC.
Discussion: UST is a human interleukin 12/23 antagonist that is highly effective for IBD and carries a low risk of malignancy and infection. Only 2-3% of patients reported arthralgias. While MM is a very rare hematologic condition, there was 1 reported case during the phase 2/3 trials of UST in a patient with longstanding IBD and anti-TNF exposure. Literature reports one additional case of MM after induction therapy with UST. Our patient is the third reported case of MM in association with UST use. Further studies are needed to determine if UST could lead to increased signaling or risk for MM. More importantly, this patient will need a multidisciplinary approach with her gastroenterologist and hematologist to balance treatment for underlying malignancy and active IBD.
Disclosures:
Ethan M.. Cohen, MD1, Ayowumi A. Adekolu, MD1, Jennifer Hadam-Veverka, MD2. P2250 - Multiple Myeloma in a Patient With a Severe Ulcerative Colitis Exacerbation on Ustekinumab, ACG 2023 Annual Scientific Meeting Abstracts. Vancouver, BC, Canada: American College of Gastroenterology.
1West Virginia University School of Medicine, Morgantown, WV; 2West Virginia University, Morgantown, WV
Introduction: Inflammatory bowel disease (IBD) is a term that includes two chronic inflammatory diseases of the GI tract, Crohn's disease and ulcerative colitis (UC). Uncontrolled inflammation can lead to bleeding, abdominal pain, hospitalization, need for surgery, and malignancy. Over the last decade, several new medications called biologics and small molecules have revolutionized the course of IBD by controlling inflammation and achieving remission. These medications come with risks which can include immunosuppression, infections or even malignancies. A detailed discussion regarding the risks and benefits of therapy should be tailored for each patient prior to starting therapy. In addition, ongoing monitoring for possible medication side effects and active disease should be done regularly.
Case Description/Methods: 60-year-old female with a history of UC diagnosed in 1990 in remission and not on medical therapy was admitted for a severe acute UC flare. Her colonoscopy showed MAYO 3 inflammation and biopsies were negative for cytomegalovirus (CMV). She improved on IV steroids and was transitioned to infliximab (IFX) as an outpatient with a steroid taper. After induction with IFX, she developed severe side effects including a psoriatic rash along with significant joint pain. Given the side effects, she was switched to ustekinumab (UST). After her first infusion with UST, her joint pain worsened, and she developed limited mobility in her left hip. A CT scan showed a lytic lesion in the left femoral neck suspicious for malignancy, and a biopsy confirmed multiple myeloma (MM). She is now undergoing treatment for her multiple myeloma but will need medications to address her active UC.
Discussion: UST is a human interleukin 12/23 antagonist that is highly effective for IBD and carries a low risk of malignancy and infection. Only 2-3% of patients reported arthralgias. While MM is a very rare hematologic condition, there was 1 reported case during the phase 2/3 trials of UST in a patient with longstanding IBD and anti-TNF exposure. Literature reports one additional case of MM after induction therapy with UST. Our patient is the third reported case of MM in association with UST use. Further studies are needed to determine if UST could lead to increased signaling or risk for MM. More importantly, this patient will need a multidisciplinary approach with her gastroenterologist and hematologist to balance treatment for underlying malignancy and active IBD.
Disclosures:
Ethan Cohen indicated no relevant financial relationships.
Ayowumi Adekolu indicated no relevant financial relationships.
Jennifer Hadam-Veverka: Abbvie – Speakers Bureau. Bristol-Myers Squibb – Speakers Bureau.
Ethan M.. Cohen, MD1, Ayowumi A. Adekolu, MD1, Jennifer Hadam-Veverka, MD2. P2250 - Multiple Myeloma in a Patient With a Severe Ulcerative Colitis Exacerbation on Ustekinumab, ACG 2023 Annual Scientific Meeting Abstracts. Vancouver, BC, Canada: American College of Gastroenterology.

